The Next Generation of Obesity Medications: What’s Coming in 2026–2027
Obesity pharmacotherapy is evolving at an unprecedented pace. Over the last several years, medications targeting the incretin hormone system—particularly GLP-1 receptor agonists—have transformed obesity treatment. Drugs such as semaglutide and tirzepatide have demonstrated weight loss approaching that seen with bariatric surgery in some patients.
The next wave of therapies is already on the horizon. Several new medications—some available in 2026 and others expected in 2027—aim to improve efficacy, convenience, and metabolic outcomes. These therapies increasingly target multiple hormonal pathways involved in appetite regulation, satiety signaling, and energy expenditure.
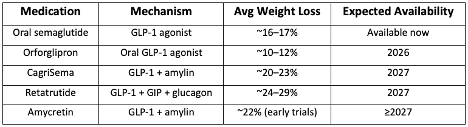
Below is an overview of the most important obesity medications expected to become available between 2026 and 2027, including their mechanism of action, expected availability, and weight-loss results from clinical trials.
1. Oral Semaglutide (Wegovy Pill)
Mechanism of Action
Semaglutide is a GLP-1 receptor agonist. GLP-1 (glucagon-like peptide-1) is a hormone released from the gut after eating that:
Increases satiety
Slows gastric emptying
Reduces appetite via central nervous system pathways
Improves glucose regulation by increasing insulin secretion and decreasing glucagon
Until recently, semaglutide for obesity required weekly injections. A new oral formulation for weight loss was approved in late 2025 and launched in early 2026. The pill uses specialized absorption technology that protects the peptide from stomach acid and enables intestinal uptake.
Clinical Trial Results
In the OASIS trials, high-dose oral semaglutide produced:
~16–17% average weight loss over about 64 weeks
Significant improvements in cardiometabolic markers such as glucose and triglycerides
These results are comparable to injectable semaglutide.
Expected Availability
Available now
This medication is significant because it provides the first oral GLP-1 therapy specifically approved for obesity.
2. Orforglipron (Eli Lilly)
Mechanism of Action
Orforglipron is a non-peptide GLP-1 receptor agonist taken orally once daily.
Unlike peptide-based GLP-1 drugs (semaglutide or tirzepatide), this drug is a small molecule, which means:
It is easier to manufacture
It does not require injection
It does not require fasting before dosing, unlike oral semaglutide
The mechanism mirrors other GLP-1 drugs:
Appetite suppression
Slower gastric emptying
Improved insulin sensitivity and glycemic control
Clinical Trial Results
Phase 3 trials have demonstrated:
~10–12.4% mean weight loss over ~72 weeks at the highest dose
Improvements in blood glucose, cholesterol, and blood pressure
These outcomes are somewhat lower than injectable incretin therapies but may be offset by greater convenience.
Expected Availability
FDA submission: 2025
Possible approval: 2026
Because it is a small-molecule drug that can be mass-produced more easily than peptide injectables, orforglipron may eventually become a more scalable obesity therapy worldwide.
3. CagriSema (Cagrilintide + Semaglutide)
Mechanism of Action
CagriSema combines two hormones that regulate appetite:
Semaglutide (GLP-1 receptor agonist)
Suppresses appetite
Slows gastric emptying
Improves insulin secretion
Cagrilintide (amylin analog)
Mimics amylin, a hormone released with insulin after meals
Promotes satiety
Reduces food intake
By targeting two complementary satiety pathways, the combination may produce greater weight loss than GLP-1 therapy alone.
Clinical Trial Results
Phase 3 REDEFINE trials demonstrated:
~20–23% average weight loss after 68 weeks
More than 40% of participants losing ≥25% of body weight
This magnitude of weight loss approaches outcomes typically seen with metabolic surgery.
Expected Availability
FDA submission: December 2025
Potential approval: late 2026 or early 2027
Estimated launch: 2027
If approved, CagriSema could become one of the most potent obesity medications available.
4. Retatrutide (Triple Hormone Agonist)
Mechanism of Action
Retatrutide represents the next major evolution in obesity pharmacotherapy. It is a triple receptor agonist targeting:
GLP-1 receptors – appetite suppression and improved glucose control
GIP receptors – enhanced insulin secretion and metabolic signaling
Glucagon receptors – increased energy expenditure and fat metabolism
This multi-pathway approach addresses obesity through both reduced caloric intake and increased metabolic expenditure.
Clinical Trial Results
Results from the TRIUMPH clinical program have been striking:
Up to ~28–29% mean weight loss at 68 weeks in late-stage trials
Earlier studies showed ~24% weight loss at 48 weeks
These outcomes are among the highest ever reported in pharmacologic obesity trials.
Expected Availability
Phase 3 trials ongoing through 2026
Possible FDA submission: late 2026 or early 2027
Potential approval: 2027
If approved, retatrutide could set a new benchmark for medical weight loss approaching bariatric surgery outcomes.
5. Amycretin (GLP-1 + Amylin Dual Agonist)
Mechanism of Action
Amycretin is an investigational drug that mimics both:
GLP-1 (appetite suppression and glucose regulation)
Amylin (satiety and slowed gastric emptying)
This dual-hormone approach resembles CagriSema but uses a single molecule rather than a combination therapy.
Clinical Trial Results
Early trials have shown promising efficacy:
~22% weight loss after 36 weeks with injectable therapy
~13% weight loss after 12 weeks in early oral-formulation trials
These early results suggest amycretin could become another high-efficacy obesity therapy. (Reuters)
Expected Availability
Phase 3 trials beginning in 2026
Potential approval late 2027 or beyond
The Future of Obesity Treatment
The obesity medication landscape is rapidly shifting from single-hormone therapies to multi-target metabolic drugs. Key trends shaping the next generation of treatments include:
1. Multi-Hormone Targeting
New drugs increasingly activate two or three metabolic pathways simultaneously, leading to greater weight loss.
Examples:
GLP-1 + GIP (tirzepatide)
GLP-1 + amylin (CagriSema)
GLP-1 + GIP + glucagon (retatrutide)
2. Oral Therapies
New small-molecule drugs like orforglipron may eliminate injections and increase accessibility.
3. Weight Loss Approaching Surgery
Clinical trials are now reporting 20–30% weight loss, a range previously achievable mainly with bariatric surgery.
4. Metabolic Health Benefits
These medications improve not only weight but also:
Type 2 diabetes
Cardiovascular risk factors
Fatty liver disease
Inflammation markers
Key Takeaways
The years 2026–2027 will likely bring several major advances in obesity pharmacotherapy, including oral GLP-1 medications and highly potent multi-hormone drugs.
The most notable upcoming medications include:
Together, these therapies may redefine obesity care—transforming it from a condition with limited treatment options into one with multiple highly effective pharmacologic therapies.